Papers
publications by categories in reversed chronological order. generated by jekyll-scholar.
2024
-
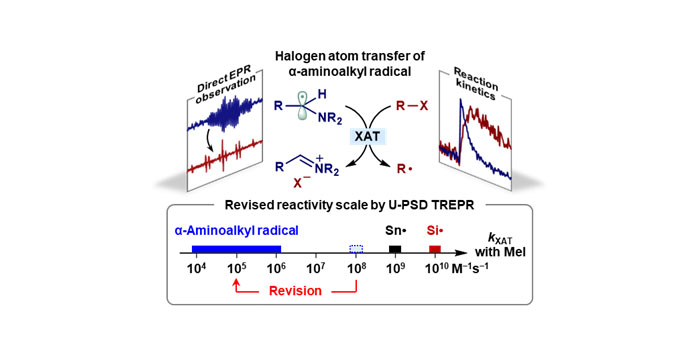 Overestimated Halogen Atom Transfer Reactivity of α-Aminoalkyl RadicalsWeiqun Suo, Jian-Qing Qi, Jing Liu, and 3 more authorsJ. Am. Chem. Soc., Sep 2024
Overestimated Halogen Atom Transfer Reactivity of α-Aminoalkyl RadicalsWeiqun Suo, Jian-Qing Qi, Jing Liu, and 3 more authorsJ. Am. Chem. Soc., Sep 2024Halogen atom transfer (XAT) is a versatile method for generating carbon radicals. Recent interest has focused on α-aminoalkyl radicals as potential XAT reagents, previously reported to exhibit reactivity comparable to tin radicals. Utilizing an advanced time-resolved EPR technique, the XAT reactions between α-aminoalkyl radicals and organic halides were examined, allowing direct observation of the process through EPR spectroscopy and analysis of radical kinetics. Second-order rate constants for these reactions were determined, with some validated using transient absorption spectroscopy. The key finding is that the reactivity of α-aminoalkyl radicals in XAT reactions is 103 to 105 times lower than that of tin and silicon radicals and only slightly higher than alkyl radicals. This challenges the belief that α-aminoalkyl radicals are as reactive as tin radicals. The study on the solvent effect indicates that the XAT reaction of α-aminoalkyl radicals does not involve a highly polarized transition state, suggesting that the kinetic polar effect in this XAT process is not as significant as previously believed. The present study provides a reliable XAT reactivity scale for α-aminoalkyl radicals, which is crucial for designing XAT reactions and understanding their mechanisms.
-
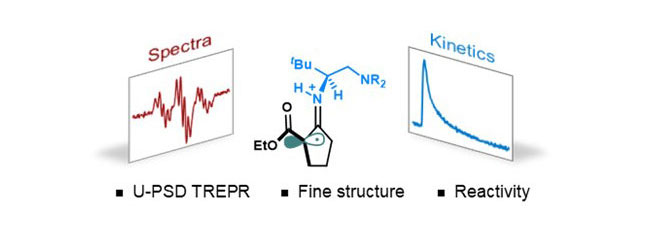 Characterization and Monitoring of Transient Enamine Radical Intermediates in Photoredox/Chiral Primary Amine Synergistic Catalytic CycleShixue Zhang, Liang Cheng, Jian-Qing Qi, and 5 more authorsCCS Chemistry, Apr 2024
Characterization and Monitoring of Transient Enamine Radical Intermediates in Photoredox/Chiral Primary Amine Synergistic Catalytic CycleShixue Zhang, Liang Cheng, Jian-Qing Qi, and 5 more authorsCCS Chemistry, Apr 2024Enamine-derived radicals are crucial intermediates in singly occupied molecular orbital (SOMO) catalysis. However, observing them directly is elusive and remains a long-standing challenge. Here, an advanced time-resolved electron paramagnetic resonance technique was employed to characterize and monitor the key intermediates in photoredox transformations by primary aminocatalysis on a microsecond timescale. The transient enamine radical cation, generated by single electron transfer (SET), and the deprotonated form of α-imino radical intermediates were directly observed for the first time, both spectroscopically and kinetically. In reactions with styrene, enamine radical cation was found to be faster than α-imino radical by one order of magnitude. This revealed the subtle role of deprotonation associated with secondary enamine radical cation in the photoredox transformations by primary aminocatalysis.
-
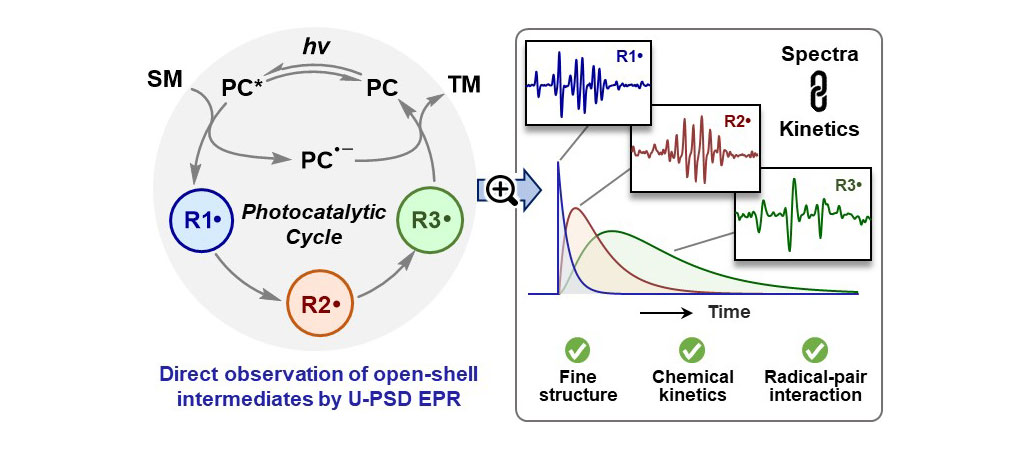 Direct observation of all open-shell intermediates in a photocatalytic cycleJian-Qing Qi, Weiqun Suo, Jing Liu, and 3 more authorsJ. Am. Chem. Soc., Mar 2024
Direct observation of all open-shell intermediates in a photocatalytic cycleJian-Qing Qi, Weiqun Suo, Jing Liu, and 3 more authorsJ. Am. Chem. Soc., Mar 2024Molecular photocatalysis has shown tremendous success in sustainable energy and chemical synthesis. However, visualizing the intricate mechanisms in photocatalysis is a significant and long-standing challenge. By employing our recently developed sensitivity-enhanced time-resolved electron paramagnetic resonance technique, we directly observed all radicals and radical ions involved in the photocatalytic addition of a tertiary amine to tert-butyl acrylate. The full picture of the photocatalytic cycle has been vividly illustrated by the fine structures, chemical kinetics, and dynamic spin polarization of all open-shell intermediates directly observed in this prototypical system. Given the universality of this methodology, we believe it greatly empowers the research paradigm of direct observation in both photocatalysis and radical chemistry.
2023
-
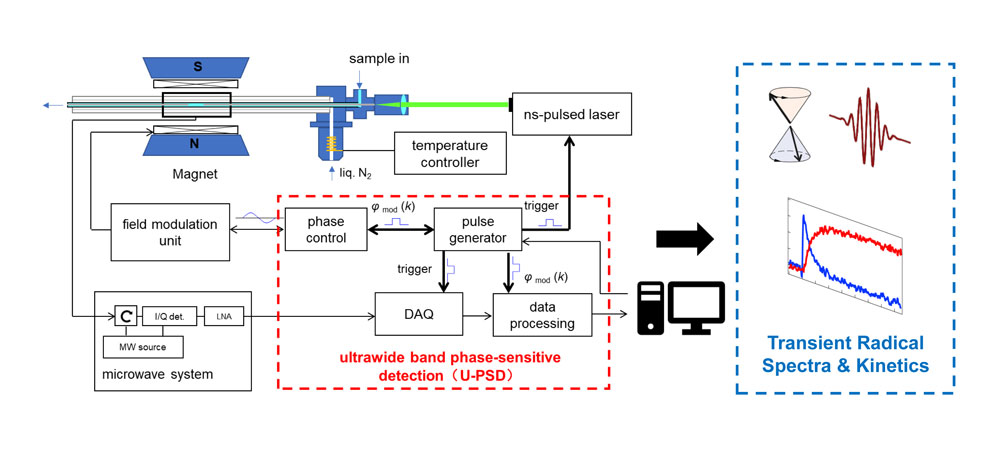 Time-resolved electron paramagnetic resonance spectrometer based on ultrawide single-sideband phase-sensitive detectionShixue Zhang, Shengqi Zhou, Jianqing Qi, and 2 more authorsRev. Sci. Instrum., Aug 2023
Time-resolved electron paramagnetic resonance spectrometer based on ultrawide single-sideband phase-sensitive detectionShixue Zhang, Shengqi Zhou, Jianqing Qi, and 2 more authorsRev. Sci. Instrum., Aug 2023A time-resolved electron paramagnetic resonance (TREPR) method with 40 ns time resolution and a high sensitivity suitable for the detection of short-lived radicals under thermal equilibrium is developed. The key is the introduction of a new detection technique named ultrawide single sideband phase sensitive detection (U-PSD) to the conventional continuous-wave EPR, which remarkably enhanced the sensitivity for the detection of broadband transient signals compared with the direct detection protocol. By repeatedly triggering a transient kinetic event f(t) (e.g., by laser flash photolysis) under a 100 kHz magnetic field modulation with precise phase control, this technique can build an ultrawide single sideband modulated signal. After single sideband demodulation, the flicker noise-suppressed signal f(t) with wide bandwidth is recovered. A U-PSD TREPR spectrometer prototype has been built, which integrated timing sequence control, laser flash excitation, data acquisition systems, and the U-PSD algorithm with a conventional continuous-wave EPR. It exhibited excellent performance in monitoring a model transient radical system, laser flash photolysis of benzophenone in isopropanol. Both the intense chemically induced dynamic electron polarization signals and the much weaker thermal equilibrium EPR signals of the generated acetone ketyl radical and benzophenone ketyl radical were clearly observed within a wide timescale ranging from sub-microsecond to milliseconds. This prototype validated the feasibility of the U-PSD technique and demonstrated its superior performance in studying complex photochemical systems containing various transient radicals, which complements the established TREPR techniques and provides a powerful tool for deep mechanistic understandings, such as in photoredox catalysis and artificial photosynthesis.
2020
-
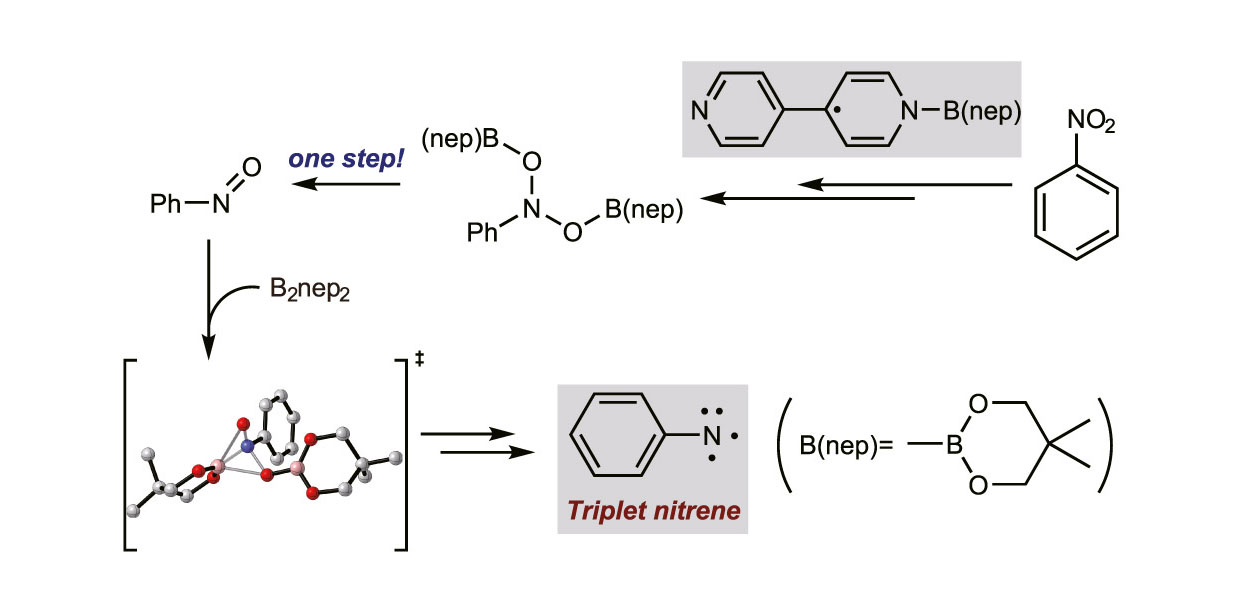 DFT Study on the Mechanism of 4,4′-Bipyridine-Catalyzed Nitrobenzene Reduction by Diboron(4) CompoundsJian-Qing Qi and Lei JiaoJ. Org. Chem., Nov 2020
DFT Study on the Mechanism of 4,4′-Bipyridine-Catalyzed Nitrobenzene Reduction by Diboron(4) CompoundsJian-Qing Qi and Lei JiaoJ. Org. Chem., Nov 2020Diboron(4) compounds serve as useful reagents for borylation, diboration, and reduction in organic synthesis. A variety of pyridine derivatives have been found capable of activating diboron(4) compounds, and different reaction mechanisms have been identified. 4,4′-Bipyridine was found to activate diboron(4) to form N,N′-diboryl-4,4′-bipyridinylidene in 2015, and very recently, it has been found that this transformation is crucial in the 4,4′-bipyridine-catalyzed reduction of nitroarenes by bis(neopentylglycolato)diboron (B2nep2), which features the formation of arylnitrene intermediates. However, the mechanism of N,N′-diboryl-4,4′-bipyridinylidene formation, as well as its role in the transformation of nitroarene to arylnitrene, remains unknown. In this work, we investigated the possible pathways of this intriguing transformation and discovered several important intermediates through density functional theory (DFT) calculations. An N-boryl 4,4′-bipyridyl radical was found to be a crucial intermediate in both the formation of N,N′-diboryl-4,4′-bipyridinylidene and the reduction of nitroarene. A type of single-step reaction with three stages, including a dissociation and two migration steps, was identified in the generation of nitrosobenzene and its reduction. Arylnitrene formation was found to occur on a triplet potential energy surface, and an intersystem crossing was found to be important for achieving a reasonable activation energy barrier for nitrene formation. We anticipate our work to provide deeper insights into the nature of this reaction that could facilitate further rational design of pyridine- and bipyridine-based catalysts.