DFT study of pyridine catalyzed reactions
My initial undergraduate research, completed during the pandemic-related lockdowns.
The Story
When the pandemic hit and kept us away from the lab, I had just joined the Jiao Group and hadn’t yet started independent wet-lab experiments. Instead of waiting it out, I decided to dive into computational chemistry to solve real-world problems from my laptop.
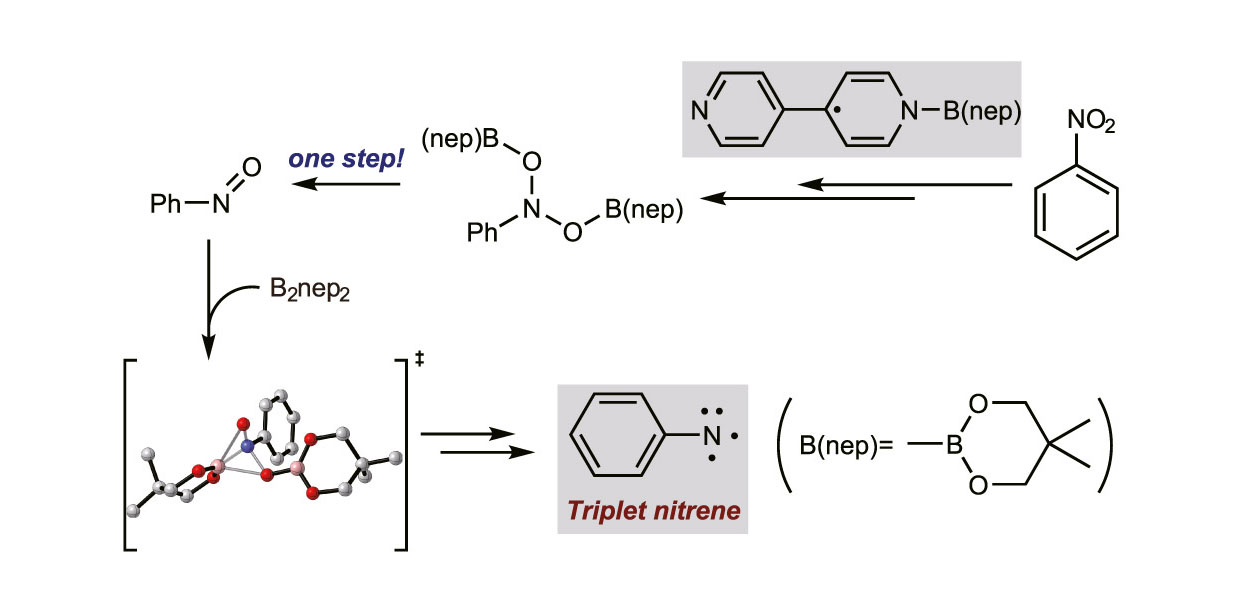
Our group has a long-standing interest in pyridine catalysis, which sparked my own curiosity. I eventually focused on the mechanism of 4,4′-Bipyridine-Catalyzed Nitrobenzene Reduction by Diboron(4) Compounds.
This period was also when I first discovered the power of programming in research. To handle the volume of data, I taught myself Python and Matlab and wrote several automation scripts to streamline my workflow.
Ultimately, I managed to resolve the reaction mechanism. With the guidance of Prof. Jiao, this work was published in The Journal of Organic Chemistry (JOC). Looking back, I feel fortunate that this challenging time turned into such a foundational experience for my career in mechanistic chemistry.
Academic Summary
This study provides a detailed mechanistic auditing of the 4,4′-bipyridine-catalyzed reduction of nitrobenzene by $\text{B}_2\text{nep}_2$ using Density Functional Theory calculations.1 The research establishes that the activation of diboron(4) compounds begins with the generation of a crucial N-boryl 4,4’-bipyridyl radical intermediate (18) through a sequence involving a [3,3]-sigmatropic rearrangement and subsequent homolytic cleavage.
[Image of a [3,3] -sigmatropic rearrangement mechanism]
This radical species then drives the formation of the boryl bipyridinylidene intermediate (3) via a radical chain mechanism. In this catalytic cycle, intermediate 3 functions as a stable reservoir that releases radical 18 to react with nitrobenzene, ultimately yielding intermediate 30.
The subsequent elimination of $(\text{Bnep})_2\text{O}$ to afford nitrosobenzene is identified as a complex, single-step reaction that proceeds through three distinct stages of dissociation and migration. A similar single-step pathway governs the interaction between nitrosobenzene and $\text{B}_2\text{nep}_2$ to form intermediate 7.
A key discovery in this work concerns the formation of arylnitrene, which is found to occur on a triplet potential energy surface. The transition from the singlet manifold to the triplet intermediate (35) via intermediate 34 requires an intersystem crossing (ISC).
[Image of an intersystem crossing Jablonski diagram]
This crossing is essential to achieving a kinetically feasible activation energy barrier for the nitrene generation process. These insights into radical chain processes and spin-multiplicity changes provide a theoretical framework for the rational design of pyridine- and bipyridine-based catalysts in reductive transformations.